
Medically integrated dispensing (MID) improves monitoring and supports value-based care, but startup costs and payer barriers remain, notes Katherine Tobon, PharmD, BCOP.

Medically integrated dispensing (MID) improves monitoring and supports value-based care, but startup costs and payer barriers remain, notes Katherine Tobon, PharmD, BCOP.

Alopecia profoundly disrupts identity, emotional wellbeing, and social life, with many people struggling due to stigma, concealment pressures, and unmet psychological support needs.

Oral semaglutide (Wegovy; Novo Nordisk) expands GLP-1 weight-loss options, but pharmacy budgets and health care cost pressures persist.

A study from Sweden, the largest of its kind, challenges the standard 24-month milestone as the key point when early relapse is a concern.

Study links elevated serum haptoglobin to chronic spontaneous urticaria activity, suggesting it predicts treatment response and complete disease control.

Real-world ONCare data show acalabrutinib lowers new-onset HTN, CV events, and discontinuations vs ibrutinib in R/R CLL/SLL.

In a pair of lively debates at the International Stroke Conference 2026, experts from around the world discussed controversies in stroke care.

OCEANIC-STROKE shows asundexian cuts recurrent ischemic stroke risk across noncardioembolic subtypes without increasing major bleeding risk.

Susan Vadaparampil, PhD, MPH, highlights disparities in cancer outcomes and trial enrollment but underscores that linking community screening programs with cancer centers may help.

Coverage of our peer-reviewed research and news reporting in the health care and mainstream press.

Experts at ISC 2026 highlighted growing evidence that GLP-1 receptor agonists may reduce stroke risk and support brain health through anti-inflammatory, vascular, and neuroprotective mechanisms.

ED dizziness visits see rising MRI use at discharge; exam-guided imaging helps detect missed strokes and informs secondary prevention.

Stroke recovery improves when care addresses social needs like reducing loneliness and depression and strengthening support.

First announced in September 2025, the online platform is set to offer access to prescription medications at steep discounts, with potential savings in the millions.

Targeted therapies, bispecifics, and CAR T-cell therapies are giving patients with multiple myeloma hope for long-term remission or a potential cure.

Across 39 patients in 2 cohorts, rapid microbe reduction, high wound closure, restored warmth and sensation, and avoided amputations were observed.
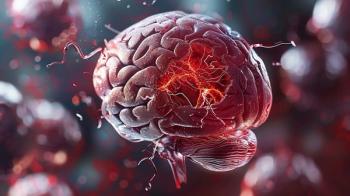
OCEANIC-STROKE shows asundexian cuts ischemic stroke recurrence by 26% in patients taking standard antiplatelet therapy, with no major bleeding increase.

A deep-learning serial CT response score outperformed conventional imaging measures in predicting OS for patients with advanced NSCLC treated with ICIs.

City St. George’s researchers identified 9 genetic signatures and epigenetic changes driving lapatinib resistance in HER2-positive breast cancer.

Oral Wegovy shifts GLP-1 obesity costs: insurance may match injectables, while cash-pay and pharmacy platforms unlock lower prices and aid programs.

The FTC first sued Express Scripts, CVS Caremark, and Optum in 2024, claiming anticompetitive and unfair rebating practices surrounding insulin.

Experts propose a 6-parameter framework for ATTR-CM monitoring and disease progression in transthyretin amyloid cardiomyopathy.

Yearly dental visits were associated with fewer secondary strokes, and hyperthyroidism was flagged as a key CVST risk factor in women.

Shervin Badihian, MD, explains how digital health tools can help boost poststroke activity via reminders, goals, feedback, and gamification.

Collaborative care use has surged 26-fold, but state reimbursement gaps leave many patients without integrated mental health care in primary care.

Rising stroke rates in young adults highlight the need to manage blood pressure and cholesterol early and improve outcomes through better ICU care.

Deaf patients face major gaps in stroke care, from poor ASL health literacy to interpreter barriers, undermining discharge understanding and emergency response.

Ateequllah Hayat, PhD, discusses epigenetic rewiring, biomarkers, and emerging strategies to overcome lapatinib resistance in HER2+ breast cancer.

CHOICE2 found intraarterial alteplase after thrombectomy raised the odds of excellent outcomes following large-vessel occlusion ischemic stroke.

Finance Committee leaders propose expanding Medicare negotiation, curbing middlemen, and reinvesting in biomedical innovation as part of a 2026 health care affordability strategy.

259 Prospect Plains Rd, Bldg H
Cranbury, NJ 08512
© 2025 MJH Life Sciences®
All rights reserved.
